Nasal Polyps

What are Nasal Polyps?
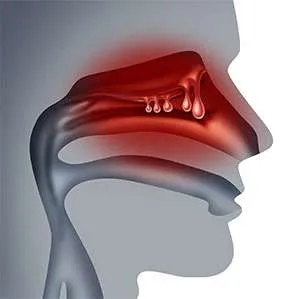
Nasal polyps are soft grape-like and sometimes teardrop-shaped growths on the lining of your nasal passageways or sinuses. They are painless and noncancerous but, if large enough can lead to difficulty in breathing, a loss of smell and sinus infections.
How Do You Get Nasal Polyps?
Nasal polyps can result from constant (chronic) inflammation of the nasal membranes, sinus infections, drug reactions, and even disorders related to your immune system. More commonly, though, they are associated with conditions that cause the nose to become inflamed, such as:
- Asthma
- Allergies
- Chronic rhinosinusitis
- Cystic fibrosis
Why we get nasal polyps is somewhat of a mystery. Some scientists believe that those who have nasal polyps indicate a different type of immune response and chemical marker in their nasal membranes than those who don’t have them.
Who’s at Risk of Getting Nasal Polyps?
Nasal polyps can form in males or females of any age; however, they’re more prominent in young to middle-aged adults. As mentioned, they can form in your nasal passageways or sinuses but most commonly appear near where your sinuses drain into your nose.
Symptoms of Nasal Polyps
It’s more common for nasal polyps to appear due to allergic rhinitis and chronic sinusitis which is an inflammation of your nasal passageways or sinuses lasting more than 12 weeks.
The most common signs of nasal polyps and chronic sinusitis are:
- Runny nose
- Nosebleeds
- Constant stuffy nose
- Chronic postnasal drip
- Loss (decreased) of sense of smell
- Loss of sense of taste
- Upper teeth pain
- Headache (or facial pressure)
- Snoring
Treatments for Nasal Polyps
 Nasal, Oral & Injectable Corticosteroids
Nasal, Oral & Injectable Corticosteroids
The typical treatment for nasal polyps is to prescribe corticosteroid nasal sprays to reduce polyp size or to remove the nasal polyps altogether. If the nasal corticosteroid isn’t completely effective, our nasal polyp doctors may follow-up with oral corticosteroids, or in severe cases, an injectable corticosteroid.
We have found that, in most cases, nasal polyps are not eradicated by medication and that only nasal polyps surgery will be completely effective in fully removing the polyps and their base of origin in the nose or sinuses.
Stereotactic Endoscopic Sinus Surgery
We have pioneered Endoscopic Sinus Surgery in the Delaware Valley. We not only offer a long-lasting, safe and gentle cure for removing nasal polyps but we do it with no expected bruising, swelling, or nasal packing.
When we use stereotactic image-guided surgery (IGS) in conjunction with endoscopic sinus surgery, our nasal polyp doctors are able to safely and effectively remove the polyps permanently. Similar to Endoscopic Sinus Surgery, Stereotactic Endoscopic Sinus Surgery is a minimally-invasive outpatient surgical procedure performed without the need for nasal packing, bruising or swelling which allows for a fast recovery and a return to normal activities in a day or two.
During Stereotactic Endoscopic Sinus Surgery, our nasal polyp doctors are able to map a three-dimensional view of the sinus cavity with a CAT scan of the sinus which allows us to more accurately detect nasal landmarks and provide you with a more effective surgical outcome.
SINUVA Sinus Implant – a non-surgical procedure that reduces polyps

Berger Henry nasal polyp doctors also offer a non-surgical procedure for patients who suffer from repeat nasal polyps. This procedure is performed with the SINUVA™ (mometasone furoate) Sinus Implant. SINUVA is proven to reduce polyps and nasal congestion and obstruction.
What is SINUVA?
SINUVA is a sinus implant that treats nasal polyps without surgery. During a routine office visit, we will use topical and/or local anesthesia to numb your nose and sinuses. SINUVA is then placed in the sinus cavity through the nostril.
How the SINUVA Sinus Implant Works
Its innovative design provides a 2-in-1 approach:
- It opens in the sinus cavity, and
- Releases anti-inflammatory medicine to treat nasal polyps for up to 90 days
Benefits of the SINUVA Sinus Implant (at 90 days)
- Significantly reduces nasal obstruction and congestion
- Significantly improves impaired sense of smell
The most common adverse reactions observed in a clinical trial were bronchitis, upper respiratory or middle ear infection, headache, light-headedness, asthma and nose bleed. SINUVA is indicated for adults with nasal polyps, 18 years of age and older, who have had ethmoid sinus surgery.
If you’d like to learn more about treatment of nasal polyps with the SINUVA Sinus Implant, please give us a call to schedule a consultation.
Indications
SINUVA™ Sinus Implant is a drug-releasing (mometasone furoate) implant indicated for the treatment of nasal polyps, in patients ≥ 18 years of age who have had ethmoid sinus surgery.
Important Safety Information
If you have a known hypersensitivity to the mometasone furoate drug or any of the ingredients in SINUVA, do not use SINUVA. Hypersensitivity reactions, including rash, itch, and swelling have been reported with use of steroids. If steroid effects such as Cushing Syndrome and adrenal suppression appear, consult your ENT doctor immediately.
SINUVA is made from bioabsorbable polymers designed to soften over time. As the implant softens and polyps decrease, the implant may be expelled out of the nose on its own or with actions such as sneezing or forceful nose blowing. The implant can be removed 90 days after placement or earlier at the physician’s discretion. Repeat administration of SINUVA has not been studied.
As with other endoscopic sinus procedures, there are risks associated with the insertion or removal of SINUVA. SINUVA should be inserted by physicians trained in otolaryngology. Discuss risks related to insertion or removal of SINUVA with your ENT doctor. Your ENT doctor will monitor the nasal tissue adjacent to the SINUVA Sinus Implant for any signs of bleeding, irritation, infection, or perforation. SINUVA should not be used in patients with nasal ulcers or trauma. The most common adverse reactions observed in clinical studies were bronchitis, upper respiratory or middle ear infection, headache, lightheadedness, asthma, and nose bleed.
If you experience excessive nasal bleeding, symptoms of infection or symptoms suggesting that the implant has moved, such as irritation or a choking sensation in the back of the throat, immediately contact a healthcare professional. Close monitoring is recommended if you have a change of vision or a history of increased intraocular pressure, glaucoma and/or cataracts.
You may report side effects to your physician or to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. You may also report side effects to Intersect ENT at 1-866-531-6004. RX Only. For important risk and use information about SINUVA, please see Full Prescribing Information or visit www.SINUVA.com.
At Berger Henry ENT, our team of experts will guide you through this process so that you can make informed decisions about your health and comfort.
It can be a worsening challenge to leave nasal polyps untreated. At Berger Henry ENT, we’re committed to providing personalized, quality care. Don’t let nasal polyps hold you back. Contact us today to schedule a consultation and breathe your way to a better quality of life.
Book Appointment